GHLF/50-SN activity at FDA Arthritis Advisory Committee Meeting July 12th & July 13th
GHLF/50-SN activity at FDA Arthritis Advisory Committee Meeting July 12th & July 13th
On July 12th and 13th the Arthritis Advisory Committee, or AAC, met at the FDA to participate in a hearing about two potential biosimilar drugs. GHLF provided commentary to speak on behalf of the 50-State Network.
What is the AAC?
The AAC, created by the FDA, reviews and evaluates data concerning the safety and efficacy of medications and treatments for arthritis and related diseases. It is made up of both voting and non-voting members who represent all areas of the healthcare industry including: PhDs, rheumatologists, members of patient groups, non-voting members from the FDA itself. Each member brings their expertise to the table to help develop a full understanding of how a new medication or treatment might impact patients.
What products were they reviewing?
The drugs reviewed are proposed biosimilars to two popular biologics already on the market. On July 12th, the AAC reviewed Amgen’s ABP 501,a biosimilar to AbbVie’s Humira, one of the most widely-used arthritis medications. On the 13th, the committee reviewed Sandoz’s drug, denoted GP2015,a biosimilar to Amgen’s Enbrel—another widely prescribed medication. A biosimilar is a medication that is “highly similar” to its reference product. It’s important to note that the bioisimilars are not interchangeable, meaning insurance companies cannot medically switch a patient off their biologic to a biosimilar to save on cost.
How does the patient community participate?
 The FDA provides time during the hearing for many patient advocacy groups to give brief, four minute testimonies about how new biosimilars would impact patients. All present expressed a desire for more accessibility to treatment options, but were wary about safety. Seth Ginsberg, GHLF’s President, testified to make sure our community’s voice was heard.
The FDA provides time during the hearing for many patient advocacy groups to give brief, four minute testimonies about how new biosimilars would impact patients. All present expressed a desire for more accessibility to treatment options, but were wary about safety. Seth Ginsberg, GHLF’s President, testified to make sure our community’s voice was heard.
Click Here to view the Testimony PDF for ABP-501 on a separate screen
Click on the play button below to listen to Seth’s Testimony on ABP-105
Click Here to view the PDF GP2015 on a separate screen
Click the play button below to listen to Seth’s Testimony on GP2015
What the committee decided:
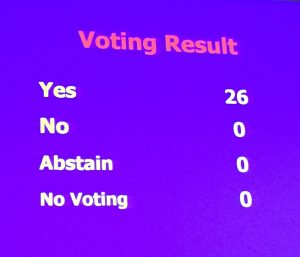 Both ABP 501 and GP2015 were unanimously approved as “highly similar” by the AAC. This approval came after the FDA and the drug companies presented their clinical and analytical data, the patient advocacy organizations gave their testimonies, and lively debate between the AAC members. The decision was made with extreme caution to ensure the best outcome for patients.
Both ABP 501 and GP2015 were unanimously approved as “highly similar” by the AAC. This approval came after the FDA and the drug companies presented their clinical and analytical data, the patient advocacy organizations gave their testimonies, and lively debate between the AAC members. The decision was made with extreme caution to ensure the best outcome for patients.
Here is what the media had to say about the AAC meeting:
