Biosimilar Legislation: Summer Update
200 million people have better access because of your advocacy
25 U.S. states and territories (as of July 2016) have passed legislation expanding access to biosimilar medications, while keeping patient safety a top priority.
This incredible milestone is a win for patients across the country suffering from chronic illnesses. Not only have over 200 million people gained a pathway to these potentially life-saving medicines, but patients in the remaining states and territories can have a renewed sense of hope, as legislation is gaining momentum across the country.
We could not have reached this milestone without your help. The unity and collaboration between patients, physicians, industry and others was able to show our elected officials the importance of this legislation in the lives of millions of people across the nation.
But we still have over half the country to go. In order to keep up our momentum, we are looking to our advocates like you to continue this call for unity and push this legislation forward.
Please see below for a list of states and territories that have already passed this legislation:
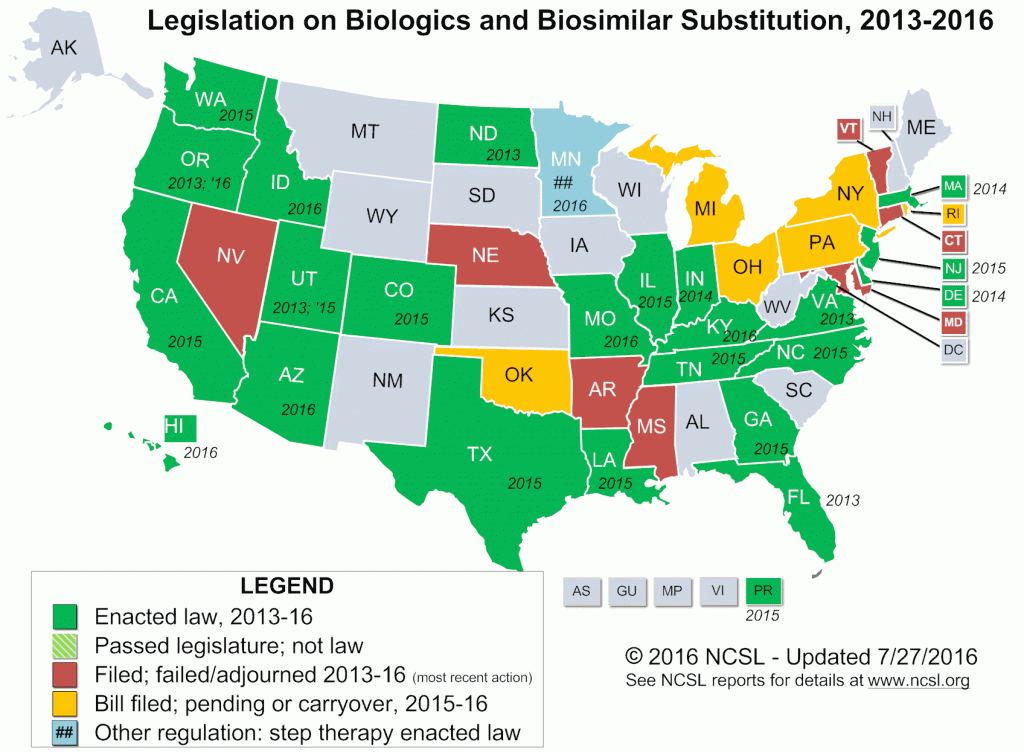
| Arizona | California | Colorado | Delaware | Georgia |
| Hawaii | Idaho | Illinois | Indiana | Kentucky |
| Louisiana | Massachusetts | Missouri | New Jersey | North Dakota |
| North Carolina | Oregon | Pennsylvania | Puerto Rico | Rhode Island |
| Tennessee | Texas | Utah | Virginia | Washington |
Is your state not on the list? Then we still need your help! Please reach out to Sarah Aoanan ([email protected]) directly if you would like to help get biosimilar legislation passed in your state.
